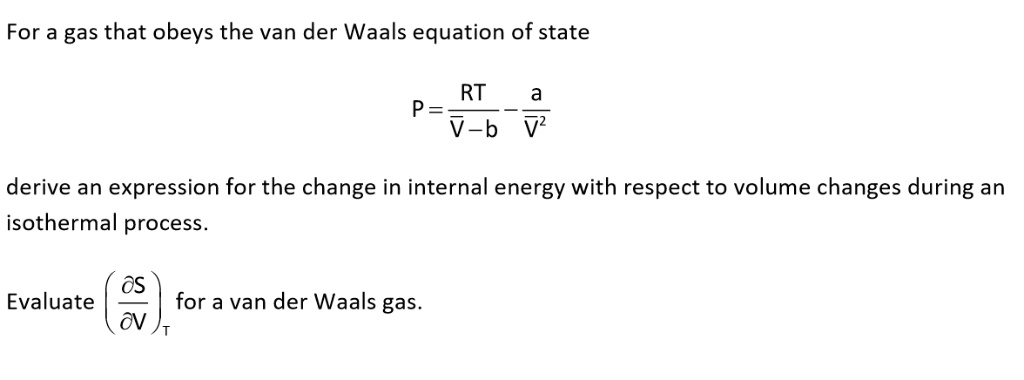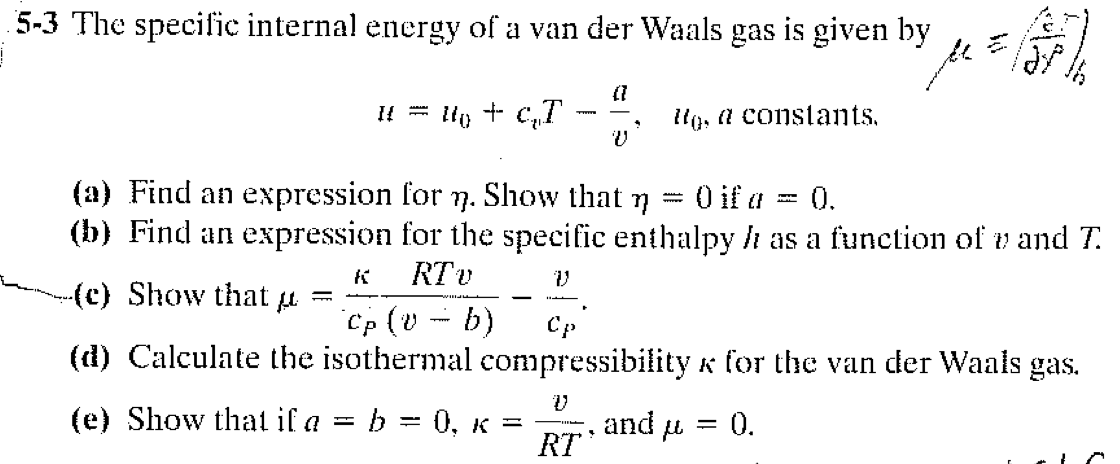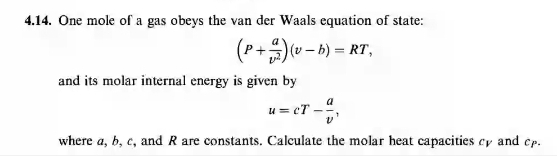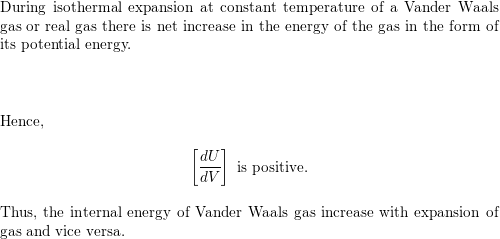SOLVED: The partition function for an interacting gas is V Nb mkg 3N/2 Z = exp ( N? a? [VkgT ) N 2th? where a and b are constants Show that the

Internal energy distribution for the ideal gas rescaled by the size K... | Download Scientific Diagram

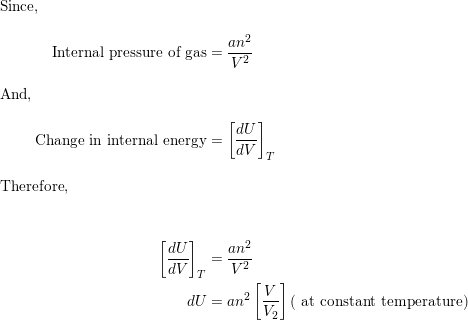
A van der Waal's gas obeys the equation of state (P+(n^(2)a)/(V^(2)))(V-nb)=nRT. Its internal energy is given by U=CT-(n^(2)a)/(V^(2)). The equation of a quasistatic adiabat for this gas is given by-
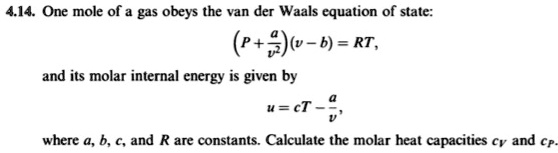
SOLVED: 414 One mole of gas obeys the van der Waals equation of state: (P+A)w b) = RT, and its molar internal energy is given by u=cT where , b, € and

OneClass: 2)Internalpressure; internal energy; partial derivatives; ideal & van derWaals gases (2...

SOLVED: Show that the internal energy departure function of van der Waals fluid can be written as: (u-pc)zx V (b) Show that the entropy departure function of van der Waals fluid can

A van der Waal's gas obeys the equation of state (P+(n^(2)a)/(V^(2)))(V-nb)=nRT. Its internal energy is given by U=CT-(n^(2)a)/(V^(2)). The equation of a quasistatic adiabat for this gas is given by-