
What is the value of log Ksp for AgI, if: Ag + I^ - → AgI + e; E^o = 0.152 V Ag → Ag^ + + e ; E^o = - 0.800 V

Regression Analysis (Evaluate Predicted Linear Equation, R-Squared, F-Test, T-Test, P-Values, Etc.) - YouTube

Calculate emf of cell at 25^(@)c Cell notation. M|underset(0.01)(M^(2+))||underset(0.0001)(M^(2+))|M if value of E(cell)^(0) is 4 volt (given (RT)/(F) in 10=0.06)

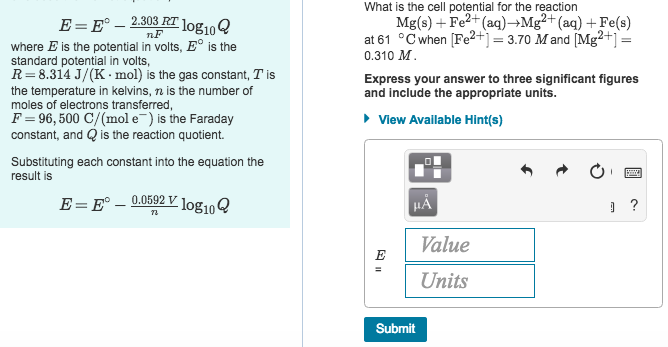
SOLVED: Part B: The Nernst Equation Write the Nernst equation? at 298 K for a redox reaction. Consider the following 11 values of reaction quotient Q 0.0100,0.0500, 0.100, 0.400, 0.700, 1.00, 10.0,

Time t ∞ Rotation of Glucose & Fructose rt r ∞ S→ G + F What id the value of k for the above reaction under given circumstances?
FIGURE. Scatter plot of RTF values for cartoon (upper panel) and motion... | Download Scientific Diagram

Given the data at 25^0 C , Ag + I^ - → AgI + e^ - ; E^0 = 0.152 V Ag → Ag^ + + e^ - ; E^0 = - 0.800 V What is the value of log Ksp for AgI ? (Given:2.303 RT /F = 0.059 V)
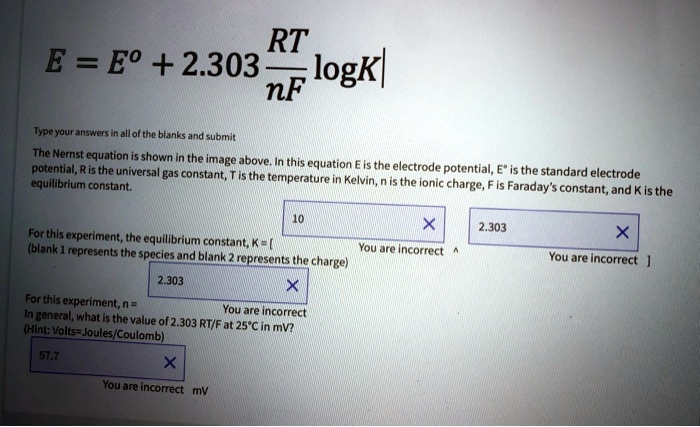
SOLVED: RT E = Eo + 2.303 logkl nF Typeyour. Kanshen allof the blanks and submit The Nernst equation shown the image above: this potential, R is the universa equation constant; is




![The emf of cell at 298 K is: [Given : 2.303 × RTF = 0.06] The emf of cell at 298 K is: [Given : 2.303 × RTF = 0.06]](https://dwes9vv9u0550.cloudfront.net/images/2198016/49d1c614-8901-47ef-8a79-1474eae4931a.jpg)

![The emf of cell at 298 K is: [Given : 2.303 × RTF = 0.06] The emf of cell at 298 K is: [Given : 2.303 × RTF = 0.06]](https://dwes9vv9u0550.cloudfront.net/images/5237783/c7440e35-87aa-4c0d-aa35-770d84a298ba.jpg)
![The emf of cell at 298 K is: [Given : 2.303 × RTF = 0.06] The emf of cell at 298 K is: [Given : 2.303 × RTF = 0.06]](https://dwes9vv9u0550.cloudfront.net/images/392604/440d00d5-9b20-4a51-a7cd-142252fa7105.jpg)




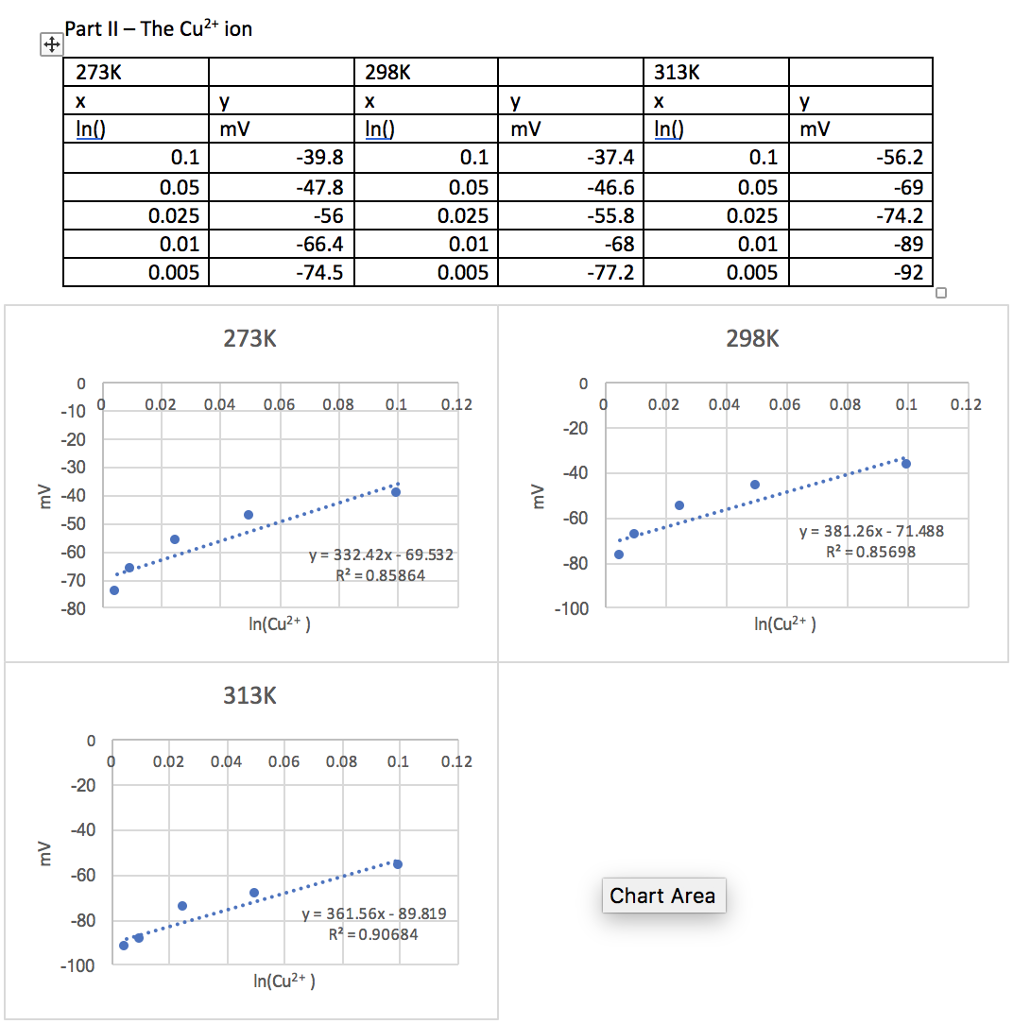


![The emf of cell at 298 K is: [Given : 2.303 × RTF = 0.06] The emf of cell at 298 K is: [Given : 2.303 × RTF = 0.06]](https://dwes9vv9u0550.cloudfront.net/images/4061150/03ae7589-d46b-44e9-9d8c-944dee23b577.jpg)

