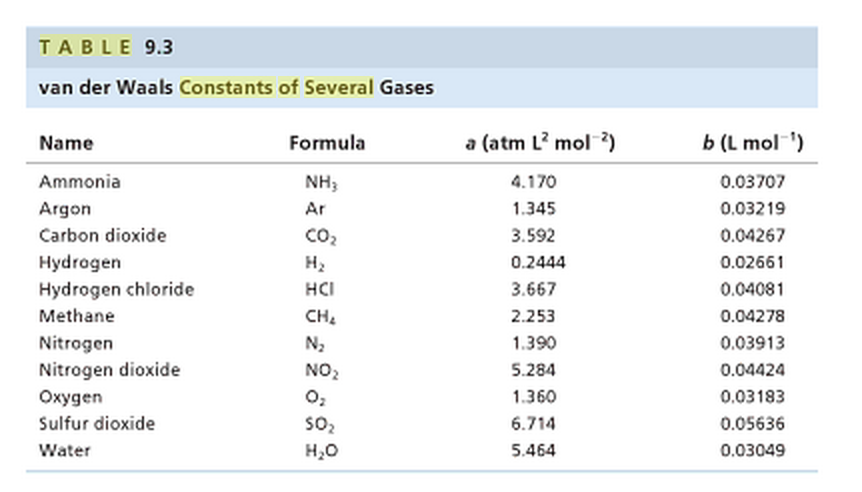
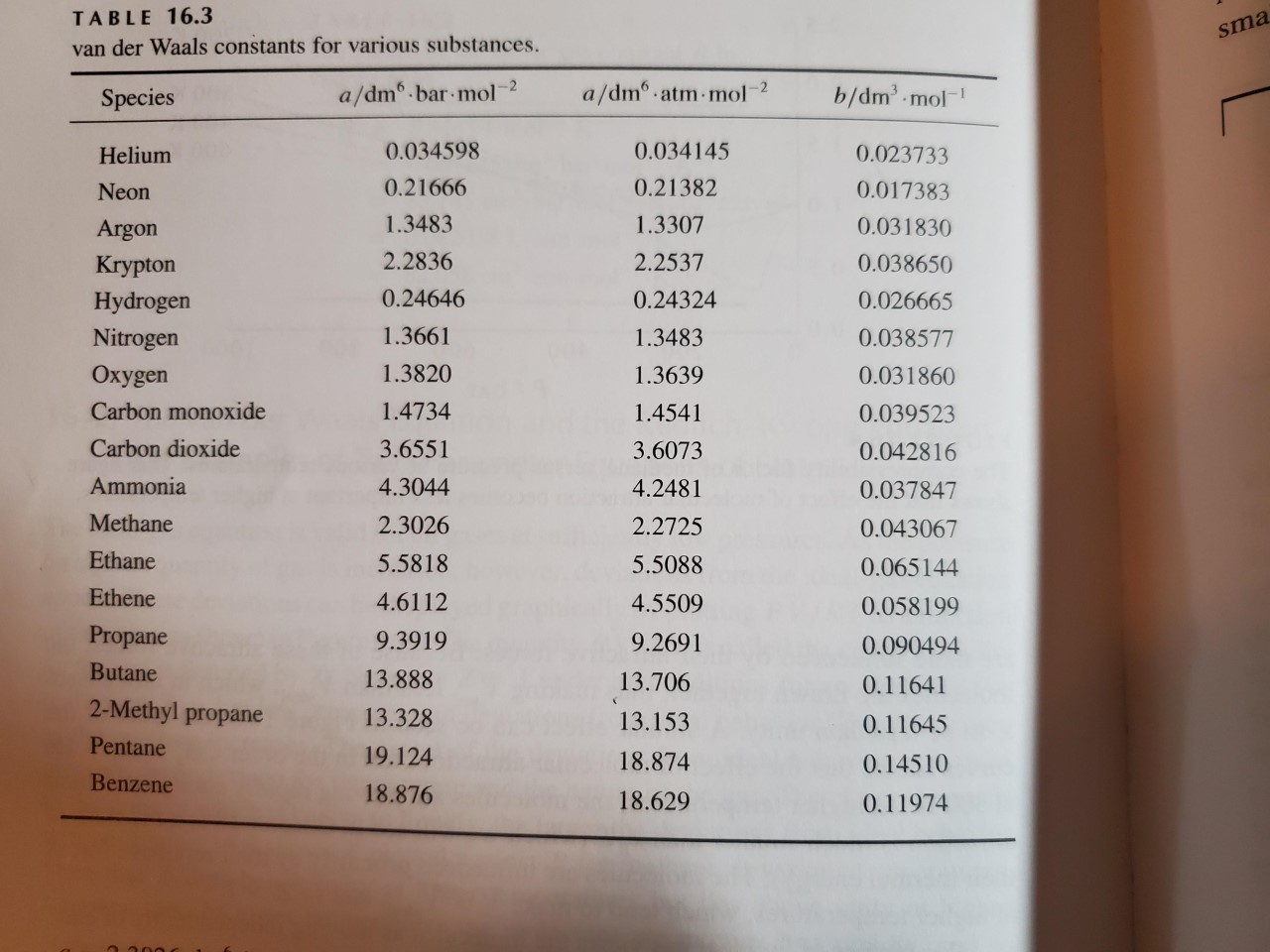
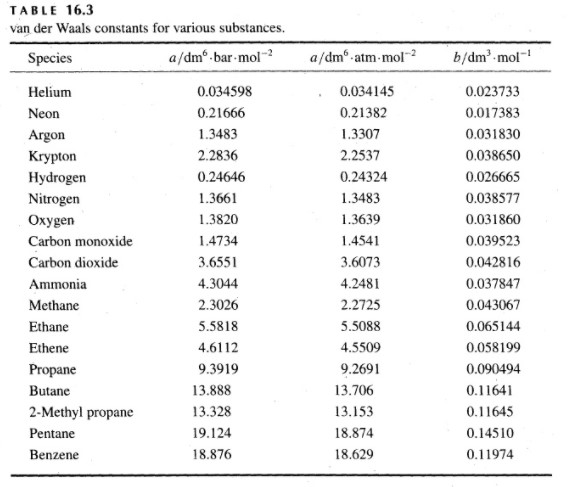
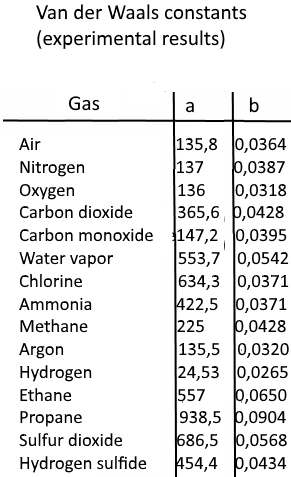
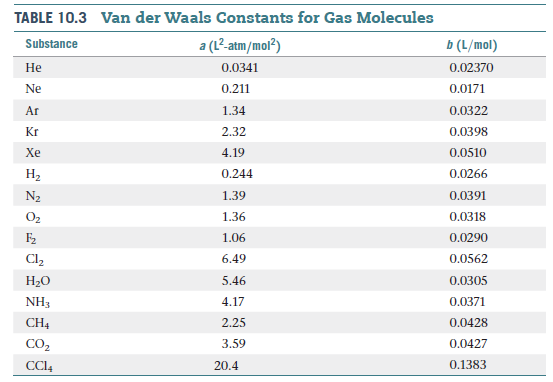
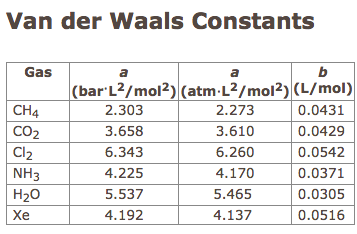
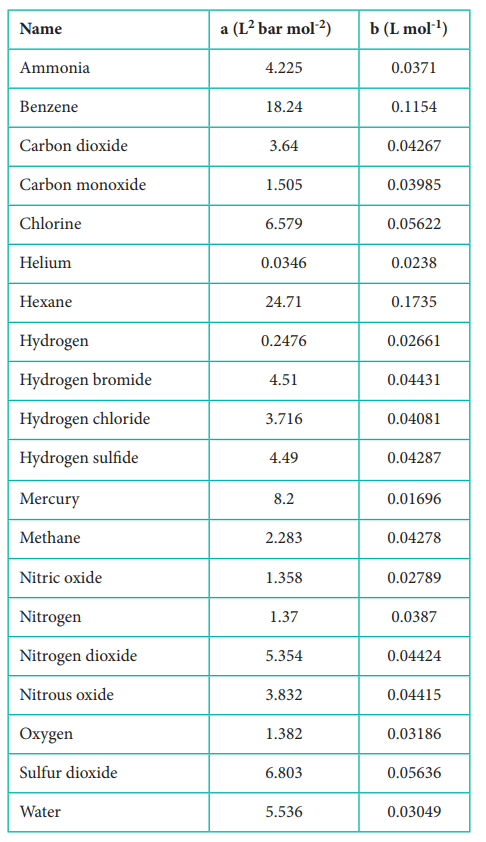
physical chemistry - Why does small value of van der Waals gas constant "b" ensure easier liquefication? - Chemistry Stack Exchange
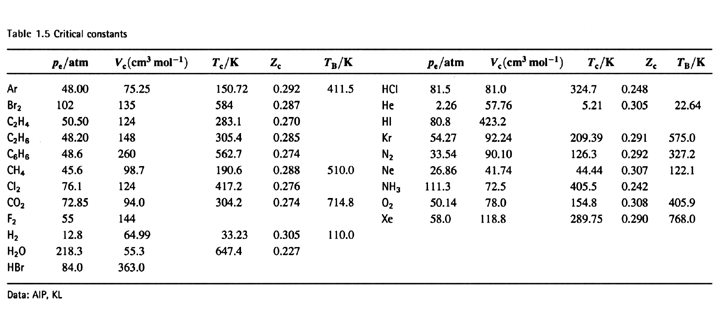
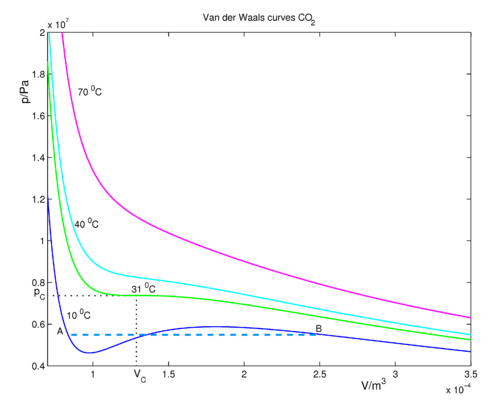
Derive the values of critical constants from the Van der Waals constants. - Sarthaks eConnect | Largest Online Education Community

The van der Waals constant for gases A, B, and C are as follows Answer the following: Which gas has the highest critical temperature?